EXHIBIT 99.1
Published on May 2, 2019

INVESTOR PRESENTATION April 2019

Forward-Looking Statements This presentation has been prepared by NeoGenomics, Inc. (“we,” ”us,” “our,” “NeoGenomics” or the “Company”) and is made for informational purposes only and does not constitute an offer to sell or a solicitation of an offer to buy securities, nor shall there be any sale of any securities in any state or jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such state or jurisdiction. The information set forth herein does not purport to be complete or to contain all of the information you may desire. Statements contained herein are made as of the date of this presentation unless stated otherwise, and neither this presentation, nor any sale of securities, shall under any circumstances create an implication that the information contained herein is correct as of any time after such date or that information will be updated or revised to reflect information that subsequently becomes available or changes occurring after the date hereof. This presentation contains “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995 relating to business, operations, and financial conditions of the Company. Words such as, but not limited to, “look forward to,” “believe,” “expect,” “anticipate,” “estimate,” “intend,” “plan,” “would,” “should” and “could,” and similar expressions or words, identify forward-looking statements. Although the Company believes the expectations reflected in such forward-looking statements are based upon reasonable assumptions, there can be no assurance that its expectations will be realized. Actual results could differ materially from those projected in the Company’s forward-looking statements due to numerous known and unknown risks and uncertainties. All forward-looking statements speak only as of the date of this presentation and are qualified in their entirety by this cautionary statement. The Company undertakes no obligation to revise or update this presentation to reflect events or circumstances after the date hereof. Non-GAAP Adjusted EBITDA "Adjusted EBITDA" is defined by NeoGenomics as net income from continuing operations before: (i) interest expense, (ii) tax expense, (iii) depreciation and amortization expense, non-cash stock-based compensation expense, and if applicable in a reporting period, acquisition-related transaction expenses (vi) non-cash impairments of intangible assets (vii) debt financing costs (viii) and other significant non-recurring or non-operating (income) or expenses. 2

Who We Are COMMON PURPOSE We save lives by improving patient care. VISION By providing uncompromising quality, exceptional service and innovative solutions, we will be the World’s leading cancer testing and information company. VALUES − Quality − Integrity − Accountability − Teamwork − Innovation We are Focused and Genuine 3

Investment Highlights Leading pure-play oncology testing company Significant market growth tailwinds Extensive molecular/oncology test menu Leader in immuno-oncology testing Market share gains driven by customer satisfaction Rapidly growing Pharma Services business Track record of profitable growth and cash flow 4

US Oncology Lab Market Clinical Reference Labs Pure Play Oncology Reference Labs Niche Oncology Players (with oncology divisions) (comprehensive test menus) (limited test menus) 5

Net Promoter Score How likely is it that you would recommend this company to a friend or colleague? (Scale of 1 to 10): • Promoters: 9-10 • Passives: 7-8 • Detractors: 0-6 • Net Promoter Score = (Promoters – Detractors) X 100 (Respondents) 500 452 Q4 2018 Clinical Client Survey 400 300 216 By The Numbers: 200 174 • Survey Period: Nov. 16th to Dec. 16th 90 100 • 1,004 respondents 24 36 4 2 1 1 4 • 15 questions + comments 0 • 2,382 comments received 10 9 8 7 6 5 4 3 2 1 0 6
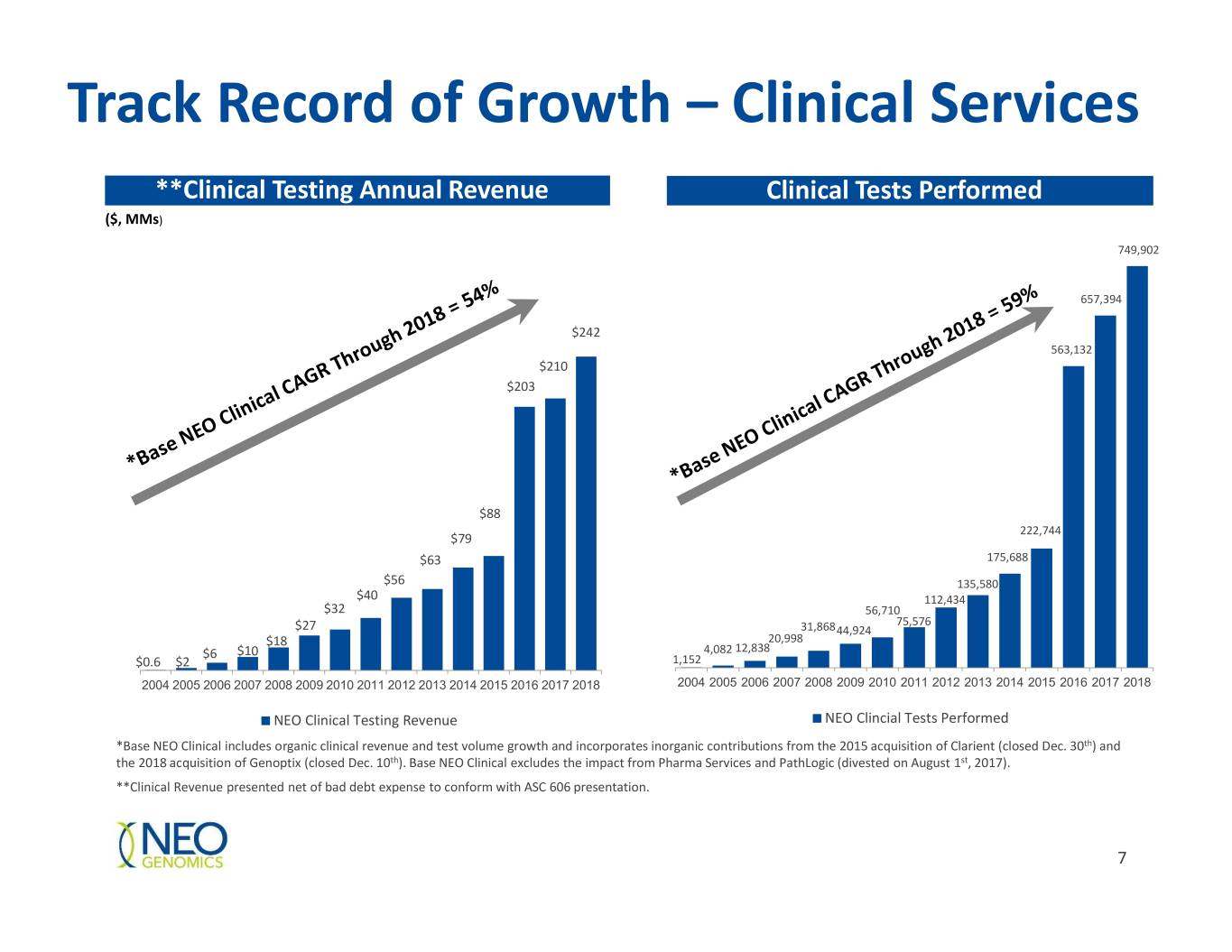
Track Record of Growth – Clinical Services **Clinical Testing Annual Revenue Clinical Tests Performed ($, MMs) 749,902 657,394 $242 563,132 $210 $203 $88 222,744 $79 $63 175,688 $56 135,580 $40 112,434 $32 56,710 75,576 $27 31,868 44,924 $18 20,998 $6 $10 4,082 12,838 $0.6 $2 1,152 2004 2005 2006 2007 2008 2009 2010 2011 2012 2013 2014 2015 2016 2017 2018 2004 2005 2006 2007 2008 2009 2010 2011 2012 2013 2014 2015 2016 2017 2018 NEO Clinical Testing Revenue NEO Clincial Tests Performed *Base NEO Clinical includes organic clinical revenue and test volume growth and incorporates inorganic contributions from the 2015 acquisition of Clarient (closed Dec. 30th) and the 2018 acquisition of Genoptix (closed Dec. 10th). Base NEO Clinical excludes the impact from Pharma Services and PathLogic (divested on August 1st, 2017). **Clinical Revenue presented net of bad debt expense to conform with ASC 606 presentation. 7
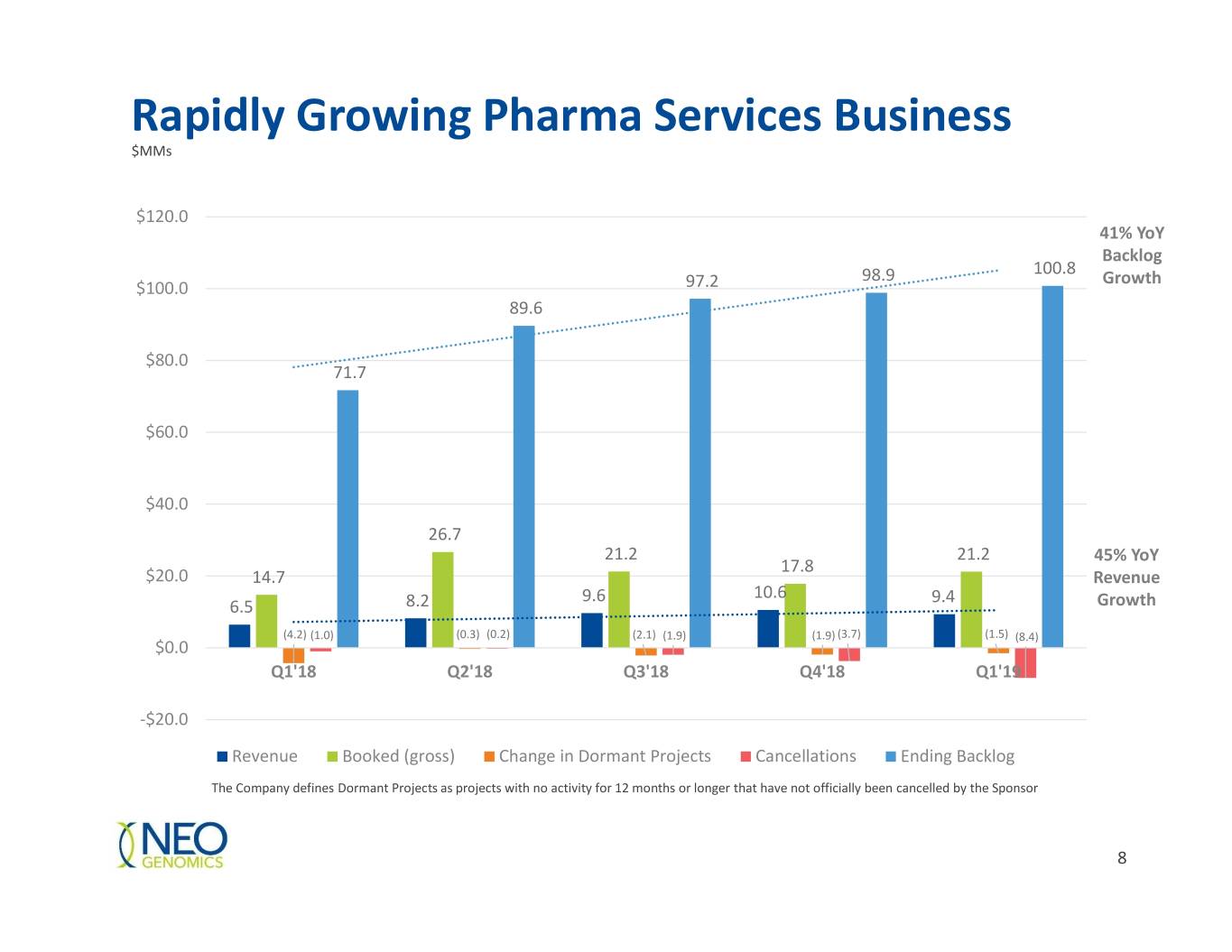
Rapidly Growing Pharma Services Business $MMs $120.0 41% YoY Backlog 100.8 98.9 Growth $100.0 97.2 89.6 $80.0 71.7 $60.0 $40.0 26.7 21.2 21.2 45% YoY 17.8 $20.0 14.7 Revenue 9.6 10.6 9.4 6.5 8.2 Growth (4.2) (1.0) (0.3) (0.2) (2.1) (1.9) (1.9) (3.7) (1.5) (8.4) $0.0 Q1'18 Q2'18 Q3'18 Q4'18 Q1'19 -$20.0 Revenue Booked (gross) Change in Dormant Projects Cancellations Ending Backlog The Company defines Dormant Projects as projects with no activity for 12 months or longer that have not officially been cancelled by the Sponsor 8
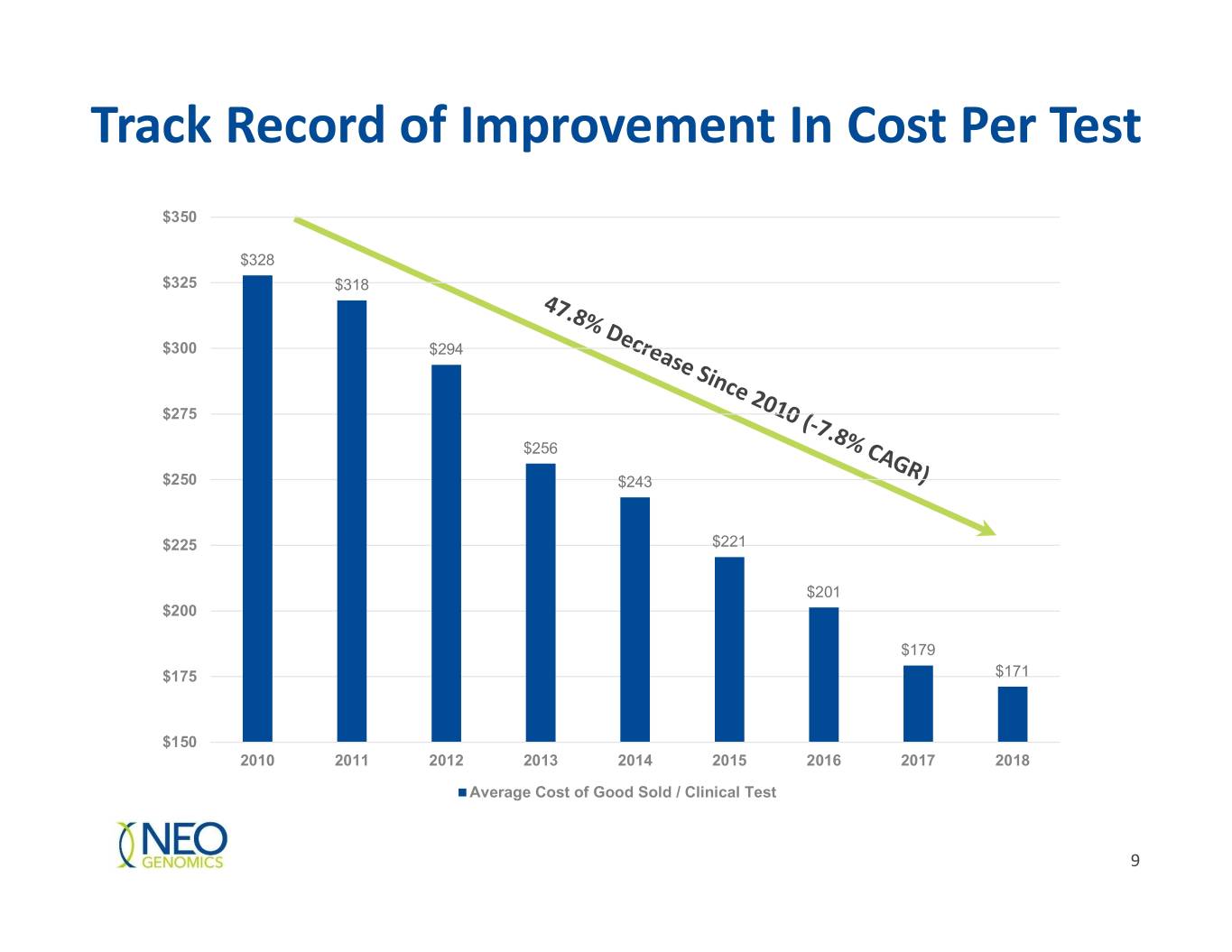
Track Record of Improvement In Cost Per Test $350 $328 $325 $318 $300 $294 $275 $256 $250 $243 $225 $221 $201 $200 $179 $175 $171 $150 2010 2011 2012 2013 2014 2015 2016 2017 2018 Average Cost of Good Sold / Clinical Test 9

A New Standard in Oncology Testing NeoGenomics Well established as a leading provider of oncology testing for pathologists and hospitals Genoptix Unprecedented reach to all customer segments Broadest and deepest test menu in the Industry Outstanding reputation Deep knowledge of community oncology practice and relationships with Gold standard consults and reports community oncologists Broad portfolio of managed care/GPO contracts Highly efficient, oncology-focused operations, medical team, and sales force 10
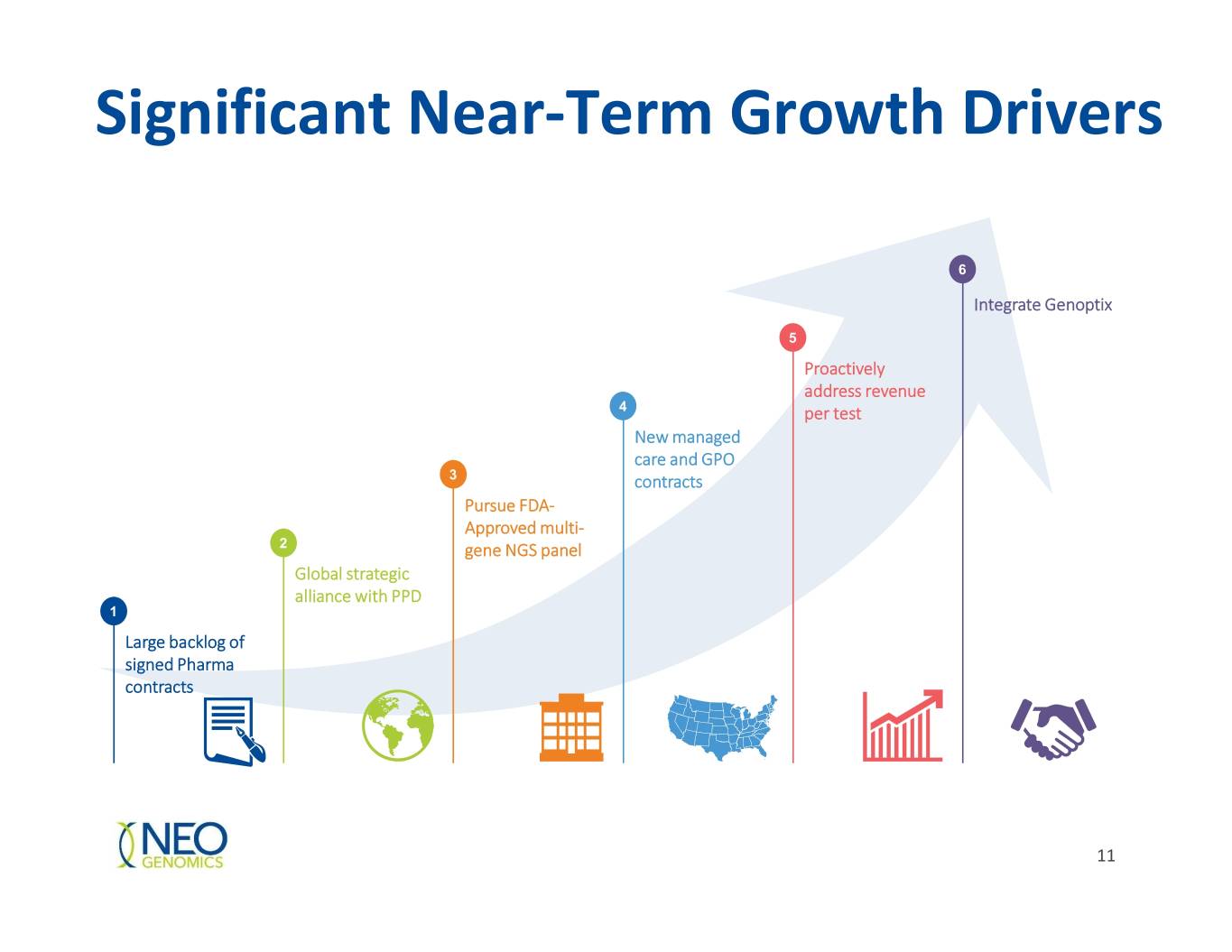
Significant Near-Term Growth Drivers 6 Integrate Genoptix 5 Proactively address revenue 4 per test New managed care and GPO 3 contracts Pursue FDA- Approved multi- 2 gene NGS panel Global strategic alliance with PPD 1 Large backlog of signed Pharma contracts 11

Oncology Market Tailwinds Aging population driving cancer incidence Increased survival driving follow-on testing Growing number of therapeutic options Increased therapeutic complexity Burgeoning oncology drug pipeline Emerging platforms and tests (NGS, TMB, MSI, etc.) 12

Appendix 13
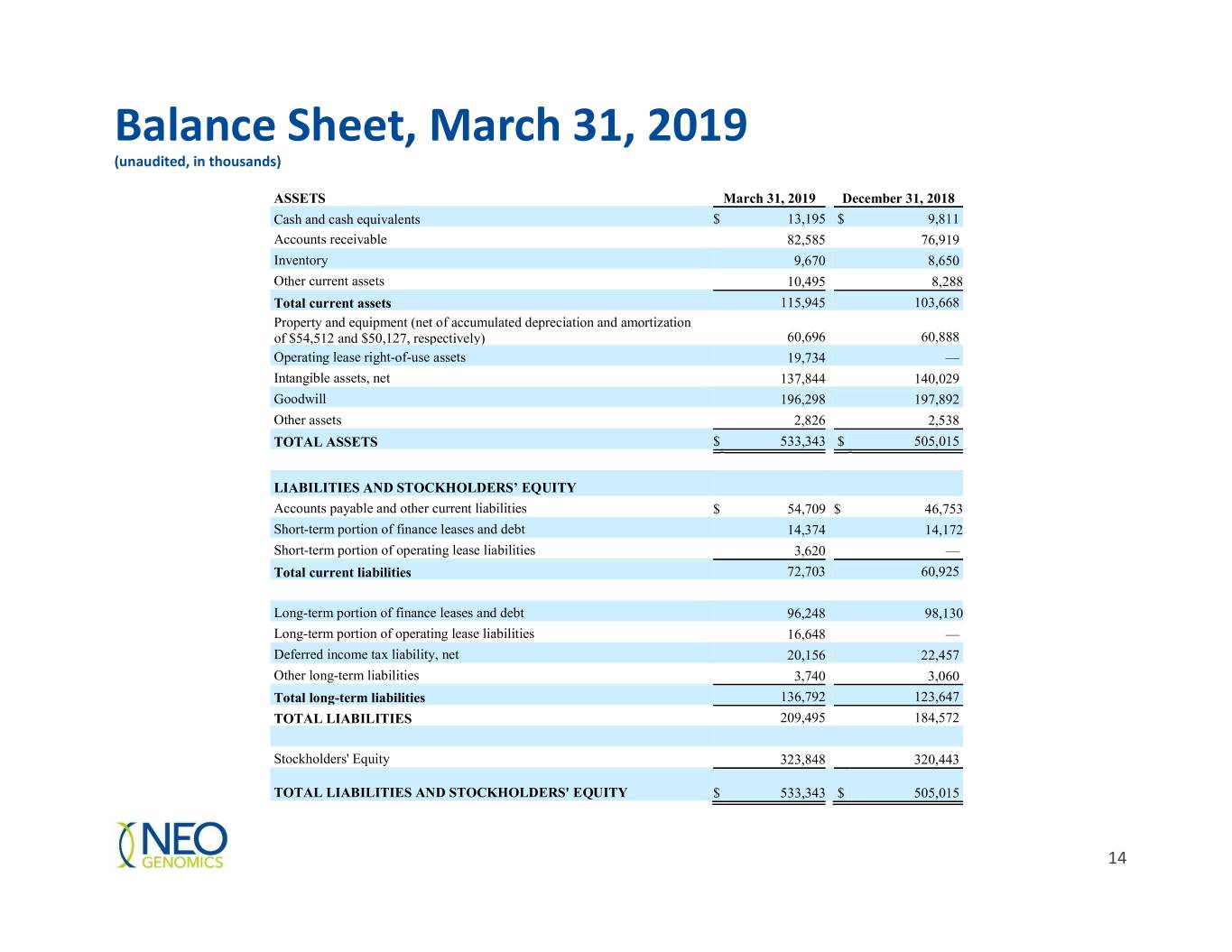
Balance Sheet, March 31, 2019 (unaudited, in thousands) ASSETS March 31, 2019 December 31, 2018 Cash and cash equivalents $ 13,195 $ 9,811 Accounts receivable 82,585 76,919 Inventory 9,670 8,650 Other current assets 10,495 8,288 Total current assets 115,945 103,668 Property and equipment (net of accumulated depreciation and amortization of $54,512 and $50,127, respectively) 60,696 60,888 Operating lease right-of-use assets 19,734 — Intangible assets, net 137,844 140,029 Goodwill 196,298 197,892 Other assets 2,826 2,538 TOTAL ASSETS $ 533,343 $ 505,015 LIABILITIES AND STOCKHOLDERS’ EQUITY Accounts payable and other current liabilities $ 54,709 $ 46,753 Short-term portion of finance leases and debt 14,374 14,172 Short-term portion of operating lease liabilities 3,620 — Total current liabilities 72,703 60,925 Long-term portion of finance leases and debt 96,248 98,130 Long-term portion of operating lease liabilities 16,648 — Deferred income tax liability, net 20,156 22,457 Other long-term liabilities 3,740 3,060 Total long-term liabilities 136,792 123,647 TOTAL LIABILITIES 209,495 184,572 Stockholders' Equity 323,848 320,443 TOTAL LIABILITIES AND STOCKHOLDERS' EQUITY $ 533,343 $ 505,015 14
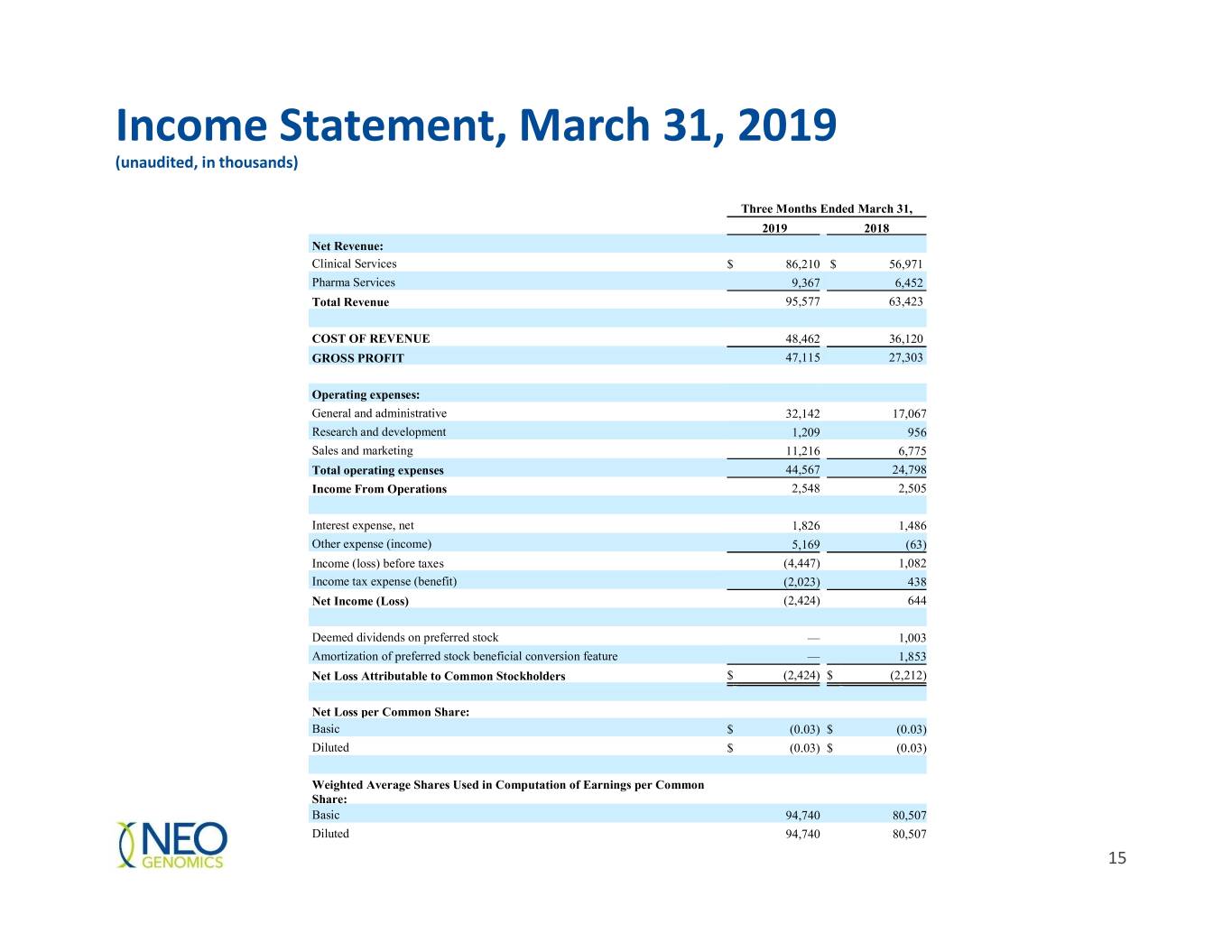
Income Statement, March 31, 2019 (unaudited, in thousands) Three Months Ended March 31, 2019 2018 Net Revenue: Clinical Services $ 86,210 $ 56,971 Pharma Services 9,367 6,452 Total Revenue 95,577 63,423 COST OF REVENUE 48,462 36,120 GROSS PROFIT 47,115 27,303 Operating expenses: General and administrative 32,142 17,067 Research and development 1,209 956 Sales and marketing 11,216 6,775 Total operating expenses 44,567 24,798 Income From Operations 2,548 2,505 Interest expense, net 1,826 1,486 Other expense (income) 5,169 (63) Income (loss) before taxes (4,447) 1,082 Income tax expense (benefit) (2,023) 438 Net Income (Loss) (2,424) 644 Deemed dividends on preferred stock — 1,003 Amortization of preferred stock beneficial conversion feature — 1,853 Net Loss Attributable to Common Stockholders $ (2,424) $ (2,212) Net Loss per Common Share: Basic $ (0.03) $ (0.03) Diluted $ (0.03) $ (0.03) Weighted Average Shares Used in Computation of Earnings per Common Share: Basic 94,740 80,507 Diluted 94,740 80,507 15

Statements of Cash Flows, March 31, 2019 (unaudited, in thousands) Three Months Ended March 31, CASH FLOWS FROM OPERATING ACTIVITIES 2019 2018 Net income (loss) $ (2,424) $ 644 Adjustments to reconcile net income to net cash provided by operating activities: Depreciation and amortization 5,271 3,633 (Gain) impairment/loss on sale of assets 156 (7) Amortization of intangibles 2,559 1,413 Amortization of debt issue costs 150 113 Stock based compensation 2,139 1,624 Changes in assets and liabilities, net (1,754) 6,892 NET CASH PROVIDED BY OPERATING ACTIVITIES 6,097 14,312 CASH FLOWS FROM INVESTING ACTIVITIES Purchases of property and equipment (3,196) (4,666) NET CASH USED IN INVESTING ACTIVITIES (3,196) (4,666) CASH FLOWS FROM FINANCING ACTIVITIES Repayment of finance lease obligations and loans (1,797) (1,394) Repayment of term loan (1,968) (6,338) Issuance of common stock, net 4,248 483 NET CASH PROVIDED BY (USED IN) FINANCING ACTIVITIES 483 (7,249) Effects of foreign exchange rate changes on cash and cash equivalents — (45) NET CHANGE IN CASH AND CASH EQUIVALENTS 3,384 2,352 CASH AND CASH EQUIVALENT, BEGINNING OF PERIOD 9,811 12,821 CASH AND CASH EQUIVALENTS, END OF PERIOD $ 13,195 $ 15,173 SUPPLEMENTAL DISCLOSURE OF CASH FLOW Interest paid $ 1,696 $ 1,396 Income taxes paid, net of refunds 8 7 SUPPLEMENTAL DISCLOSURE OF NON-CASH INVESTING AND FINANCING INFORMATION: Equipment acquired under finance lease/loan obligations $ 2,003 $ 3,355 16
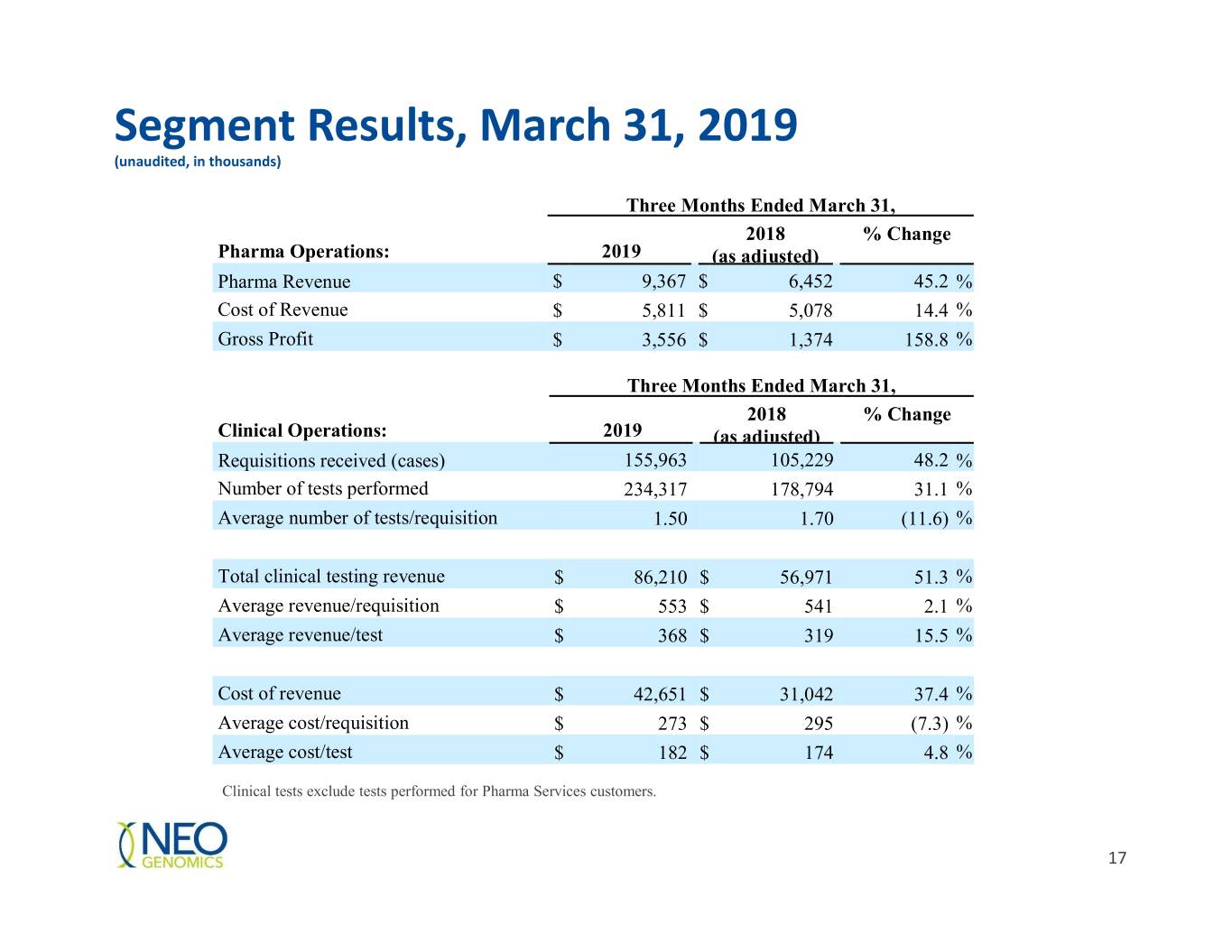
Segment Results, March 31, 2019 (unaudited, in thousands) Three Months Ended March 31, 2018 % Change Pharma Operations: 2019 (as adjusted) Pharma Revenue $ 9,367 $ 6,452 45.2 % Cost of Revenue $ 5,811 $ 5,078 14.4 % Gross Profit $ 3,556 $ 1,374 158.8 % Three Months Ended March 31, 2018 % Change Clinical Operations: 2019 (as adjusted) Requisitions received (cases) 155,963 105,229 48.2 % Number of tests performed 234,317 178,794 31.1 % Average number of tests/requisition 1.50 1.70 (11.6) % Total clinical testing revenue $ 86,210 $ 56,971 51.3 % Average revenue/requisition $ 553 $ 541 2.1 % Average revenue/test $ 368 $ 319 15.5 % Cost of revenue $ 42,651 $ 31,042 37.4 % Average cost/requisition $ 273 $ 295 (7.3) % Average cost/test $ 182 $ 174 4.8 % Clinical tests exclude tests performed for Pharma Services customers. 17
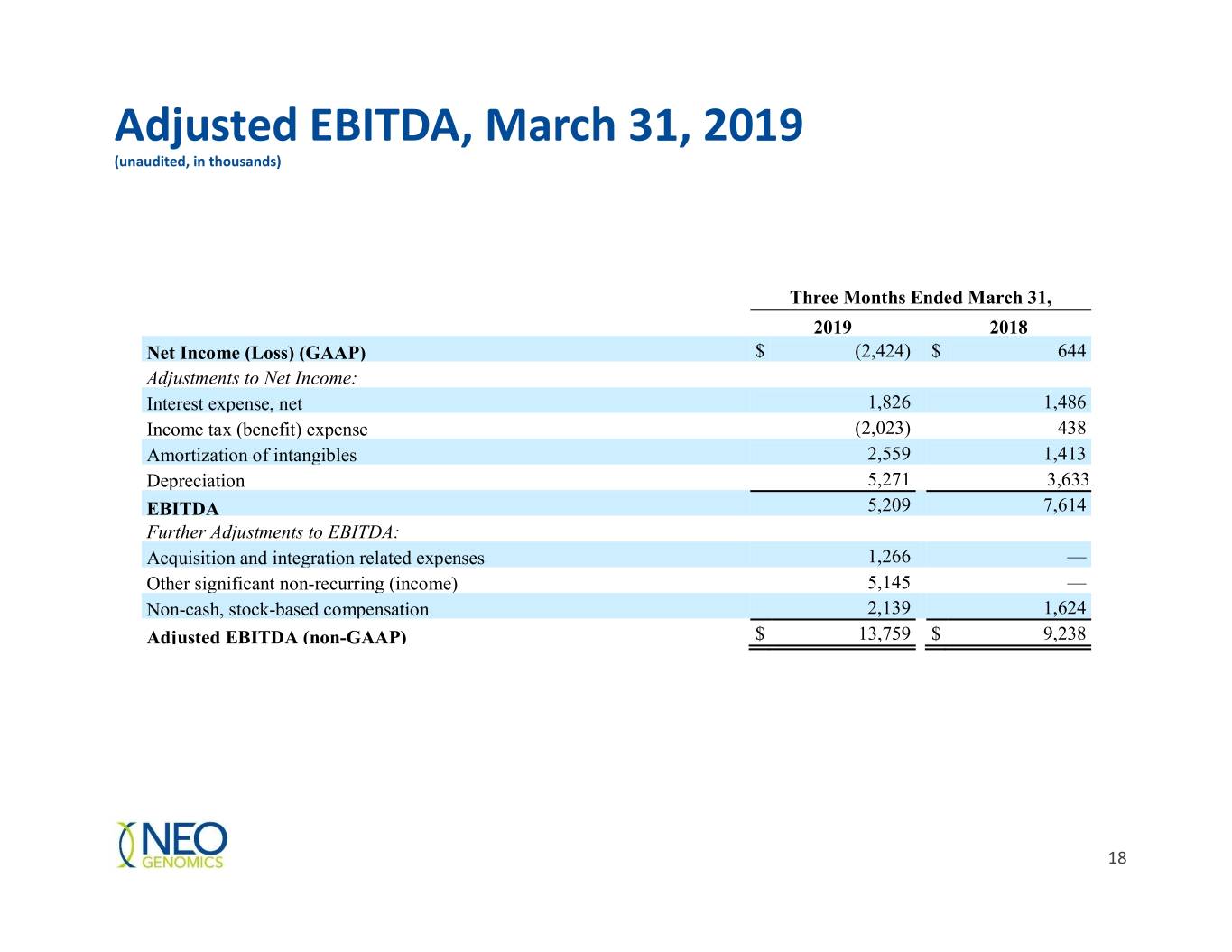
Adjusted EBITDA, March 31, 2019 (unaudited, in thousands) Three Months Ended March 31, 2019 2018 Net Income (Loss) (GAAP) $ (2,424) $ 644 Adjustments to Net Income: Interest expense, net 1,826 1,486 Income tax (benefit) expense (2,023) 438 Amortization of intangibles 2,559 1,413 Depreciation 5,271 3,633 EBITDA 5,209 7,614 Further Adjustments to EBITDA: Acquisition and integration related expenses 1,266 — Other significant non-recurring (income) 5,145 — Non-cash, stock-based compensation 2,139 1,624 Adjusted EBITDA (non-GAAP) $ 13,759 $ 9,238 18
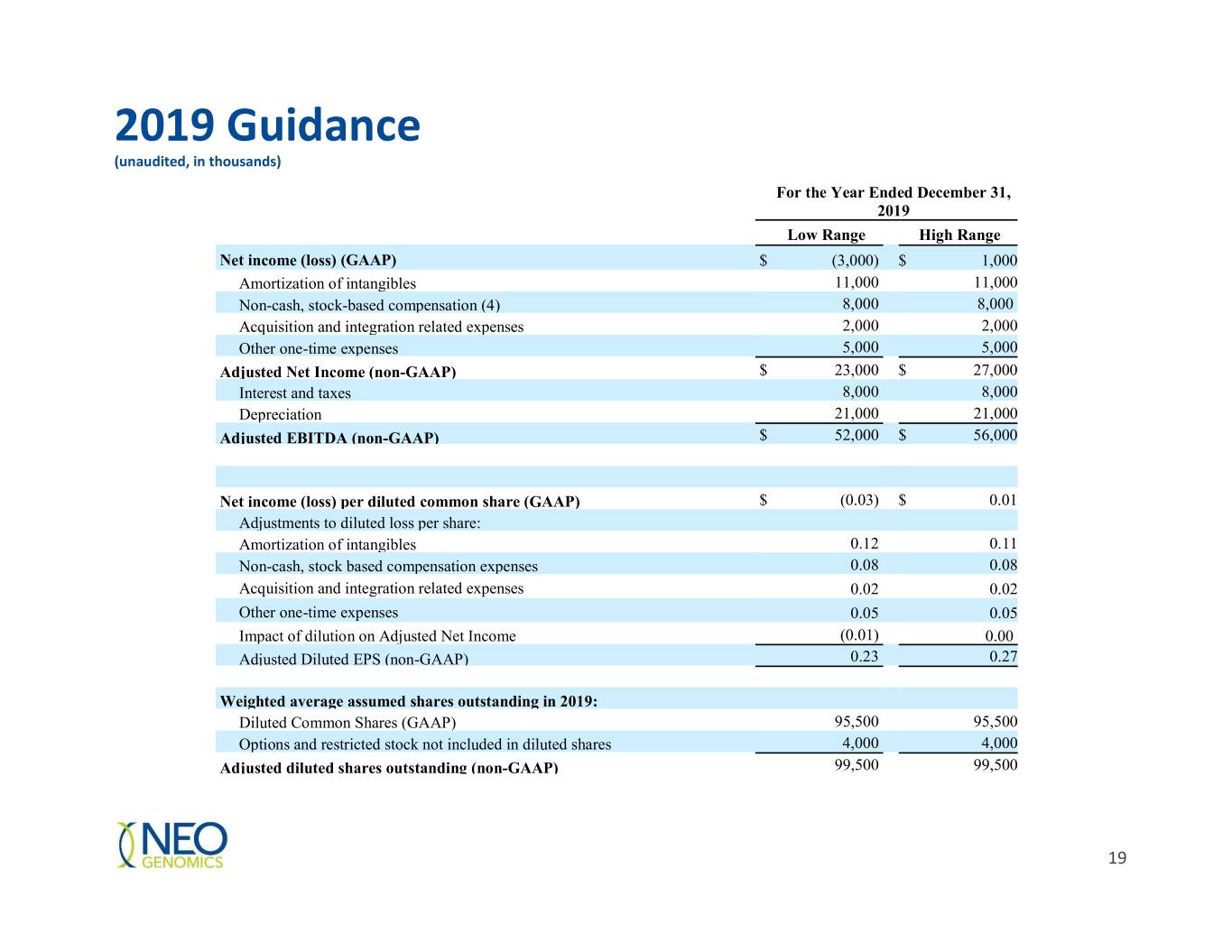
2019 Guidance (unaudited, in thousands) For the Year Ended December 31, 2019 Low Range High Range Net income (loss) (GAAP) $ (3,000) $ 1,000 Amortization of intangibles 11,000 11,000 Non-cash, stock-based compensation (4) 8,000 8,000 Acquisition and integration related expenses 2,000 2,000 Other one-time expenses 5,000 5,000 Adjusted Net Income (non-GAAP) $ 23,000 $ 27,000 Interest and taxes 8,000 8,000 Depreciation 21,000 21,000 Adjusted EBITDA (non-GAAP) $ 52,000 $ 56,000 Net income (loss) per diluted common share (GAAP) $ (0.03) $ 0.01 Adjustments to diluted loss per share: Amortization of intangibles 0.12 0.11 Non-cash, stock based compensation expenses 0.08 0.08 Acquisition and integration related expenses 0.02 0.02 Other one-time expenses 0.05 0.05 Impact of dilution on Adjusted Net Income (0.01) 0.00 Adjusted Diluted EPS (non-GAAP) 0.23 0.27 Weighted average assumed shares outstanding in 2019: Diluted Common Shares (GAAP) 95,500 95,500 Options and restricted stock not included in diluted shares 4,000 4,000 Adjusted diluted shares outstanding (non-GAAP) 99,500 99,500 19

Company Overview Comprehensive, oncology-focused test menu • Extensive molecular-oncology offering, with 150+ tests • A leader in immuno-oncology testing • In-house digital pathology expertise Extensive clinical expertise • 80+ pathologists and PhDs Global Pharma Services offering • $101 million backlog of signed contracts • Labs in US, Europe and Asia Significant scale and scope • 2600+ hospitals and cancer centers • 900,000+ tests per year • Broad geographic coverage with major labs in California, Florida and Texas 20

Our Competitive Advantage • Comprehensive, multi-modality “one-stop-shop” • Large and advanced somatic cancer test menu • Unparalleled reach into all customer segments, including hospitals, pathologists, and community oncology practices • National footprint and extensive payer contracts • Outstanding client service and partnership models • Synergistic Pharma and Clinical businesses 21

Multi-modality One-stop-shop Complementary and Comprehensive Anatomic Flow Cytometry Cytogenetics FISH Molecular Pathology • Sanger Sequencing • Extensive • Robust Library of • Consultation Pathology • State-of-Art • Next-Gen Sequencing technology automation for High Validated Probes • Immunohistochemistry • Whole Exome Quality/Low Cost • Tech-Only Service • Digital Imaging • 8-16 Color Flow Sequencing • 4-6 day TAT with Automation • Automated • MRD detection • Targeted Sequencing quantitative IHC • Becton Dickinson & • Real-time qPCR Beckman Coulter • MultiOmyx • Liquid Biopsy • Nanostring/Gene Ex 22
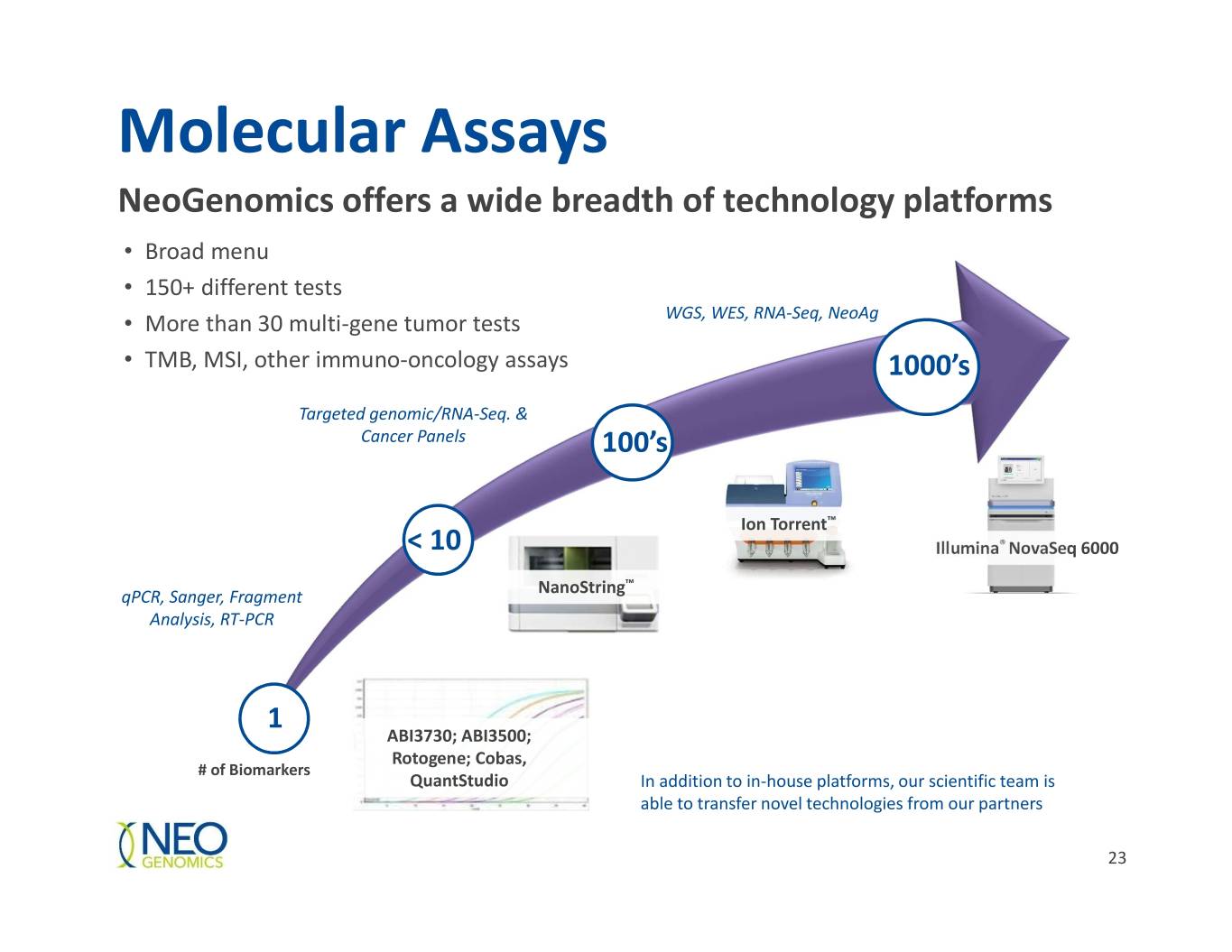
Molecular Assays NeoGenomics offers a wide breadth of technology platforms • Broad menu • 150+ different tests • More than 30 multi-gene tumor tests WGS, WES, RNA-Seq, NeoAg • TMB, MSI, other immuno-oncology assays 1000’s Targeted genomic/RNA-Seq. & Cancer Panels 100’s Ion Torrent™ < 10 Illumina® NovaSeq 6000 NanoString™ qPCR, Sanger, Fragment Analysis, RT-PCR 1 ABI3730; ABI3500; Rotogene; Cobas, # of Biomarkers QuantStudio In addition to in-house platforms, our scientific team is able to transfer novel technologies from our partners 23
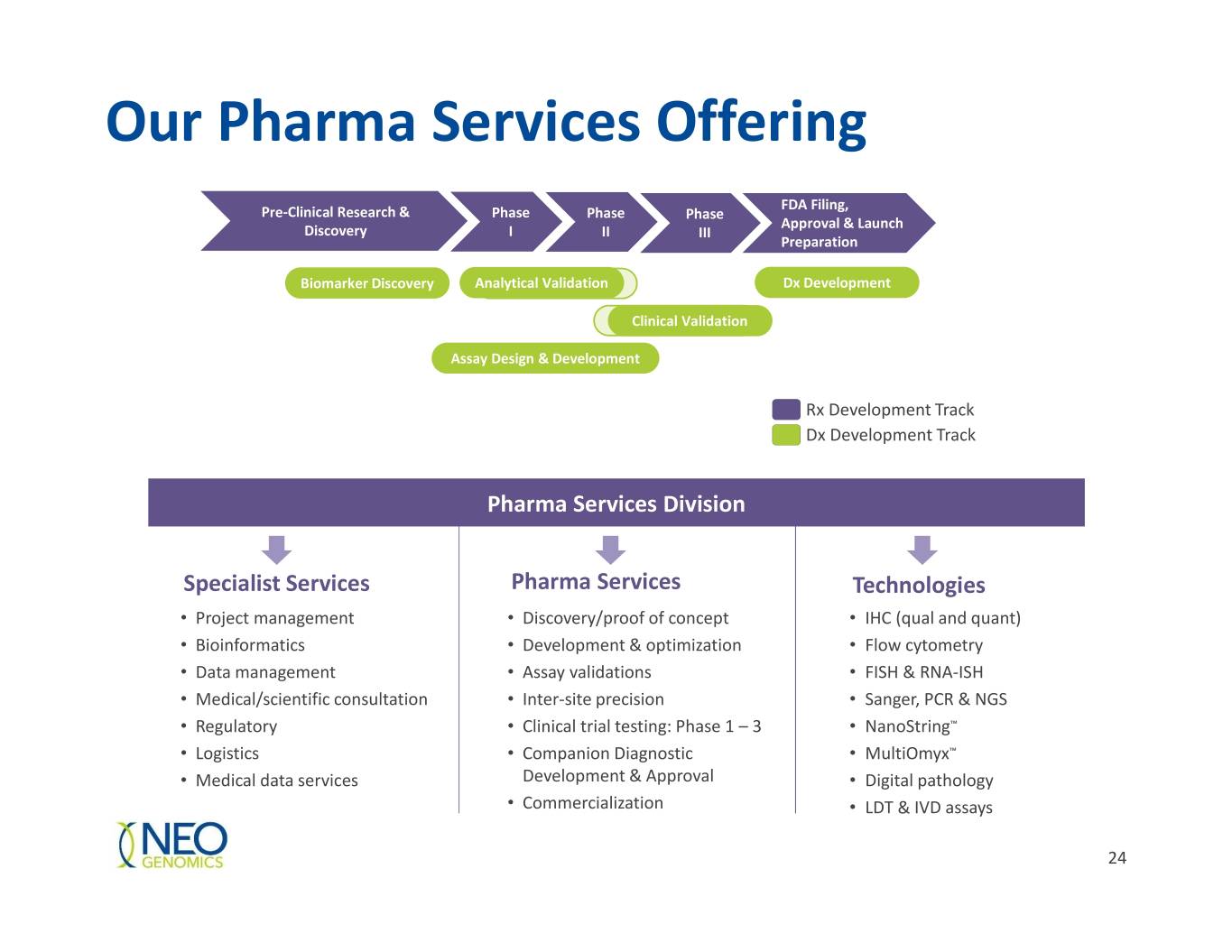
Our Pharma Services Offering FDA Filing, Pre-Clinical Research & Phase Phase Phase Approval & Launch Discovery I II III Preparation Biomarker Discovery Analytical Validation Dx Development Clinical Validation Assay Design & Development Rx Development Track Dx Development Track Pharma Services Division Specialist Services Pharma Services Technologies • Project management • Discovery/proof of concept • IHC (qual and quant) • Bioinformatics • Development & optimization • Flow cytometry • Data management • Assay validations • FISH & RNA-ISH • Medical/scientific consultation • Inter-site precision • Sanger, PCR & NGS • Regulatory • Clinical trial testing: Phase 1 – 3 • NanoString™ • Logistics • Companion Diagnostic • MultiOmyx™ • Medical data services Development & Approval • Digital pathology • Commercialization • LDT & IVD assays 24
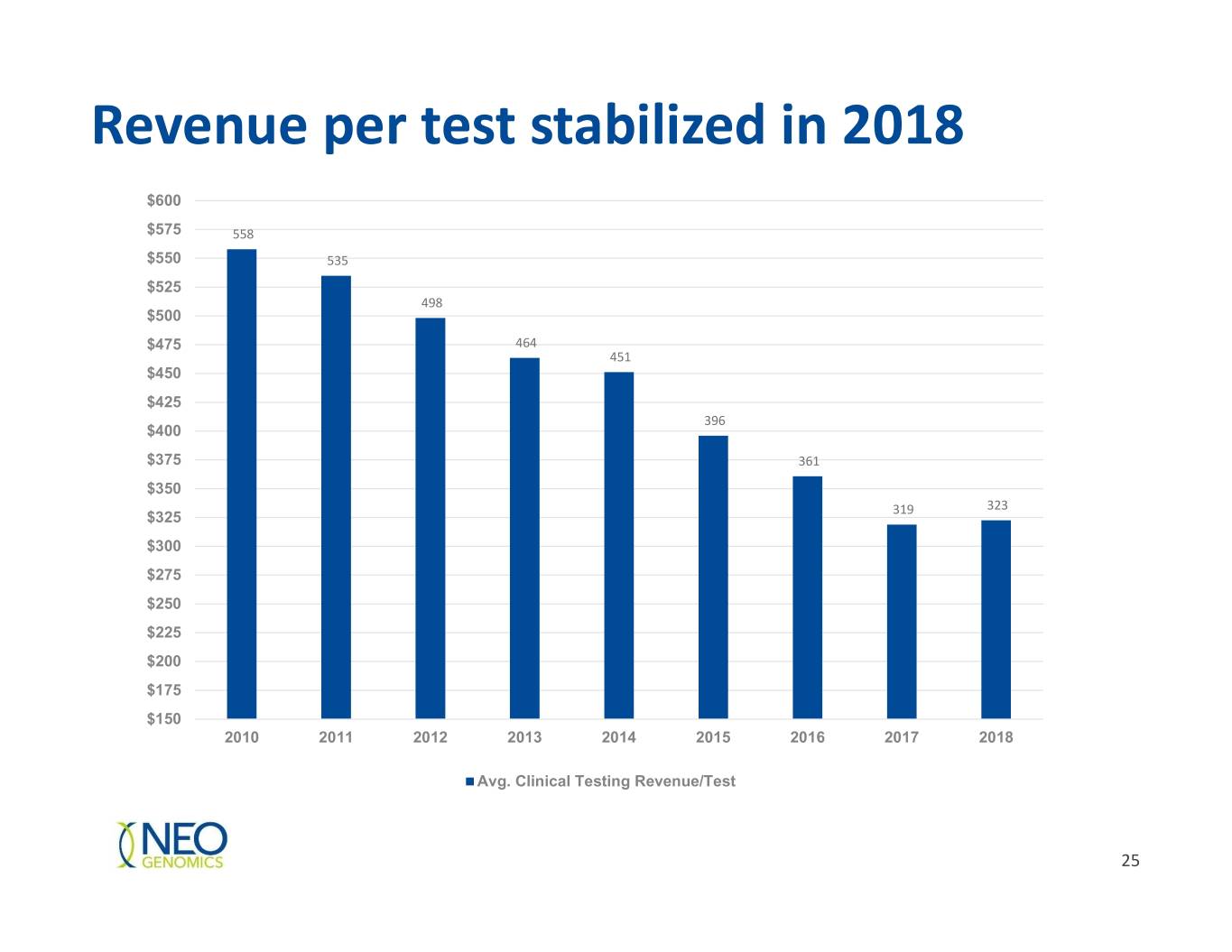
Revenue per test stabilized in 2018 $600 $575 558 $550 535 $525 498 $500 $475 464 451 $450 $425 396 $400 $375 361 $350 323 $325 319 $300 $275 $250 $225 $200 $175 $150 2010 2011 2012 2013 2014 2015 2016 2017 2018 Avg. Clinical Testing Revenue/Test 25

Global Strategic Alliance With PPD PPD’s preferred lab for oncology testing Significant revenue opportunity Expands global client base Expansion into Asia Collaborations for companion diagnostics Opportunities to leverage laboratory data for trials 26

Expands Reach into Oncology Practices Significant opportunity Genoptix is well established in for growth this market segment $2.5 billion revenue Doubles existing sales force allowing opportunity for more territories and greater penetration >2,000 independent, Customized reports specifically community oncology tailored for the oncologist practices community Important channel for fastest Specialized pathologists with years growing tests (e.g., NGS and of experience working directly with Liquid Biopsy) oncologists 27

Leverages Best Offerings MANAGED CARE CUSTOMIZED EXTENSIVE PHARMA NATIONAL AND GPO REPORTING TEST MENU SERVICES FOOTPRINT CONTRACTS NeoGenomics’ test Synergistic With operations on NeoGenomics Genoptix has a menu includes many pharma services both coasts, has a broad suite of tests that Genoptix business drives NeoGenomics will portfolio of customized does not currently be able to improve access to contracts in reporting tools offer to its customers turn-around time • Broader portfolio companion for Genoptix which that are of NGS tests diagnostics customers Genoptix does considered the • 10-color flow not participate gold standard • Greater number of among IHC tests oncologists • Solid tumor pathology 28

COMPASS® and CHART® COMPASS: comprehensive, hematopathologist-directed, integrated assessment report • Customized workflow on each patient case to provide a disease-specific evaluation based on up-to-date guidelines • Actionable diagnosis in a one-page correlation report • Consultation with assigned hematopathologist available on every case • Notification of acute cases and unexpected diagnoses within 24 hours • Real-time electronic reporting with Genoptix Online, powered by eCOMPASS™ • Review of challenging cases and presentation of tumor conferences with a Genoptix Hematopathologist through eRounds CHART: a longitudinal report including a consultative review and correlation with relevant prior findings by a Genoptix Hematopathologist, used to: • Monitor response to therapy • Determine disease progression • Evaluate clonal evolution • Assess residual disease 29
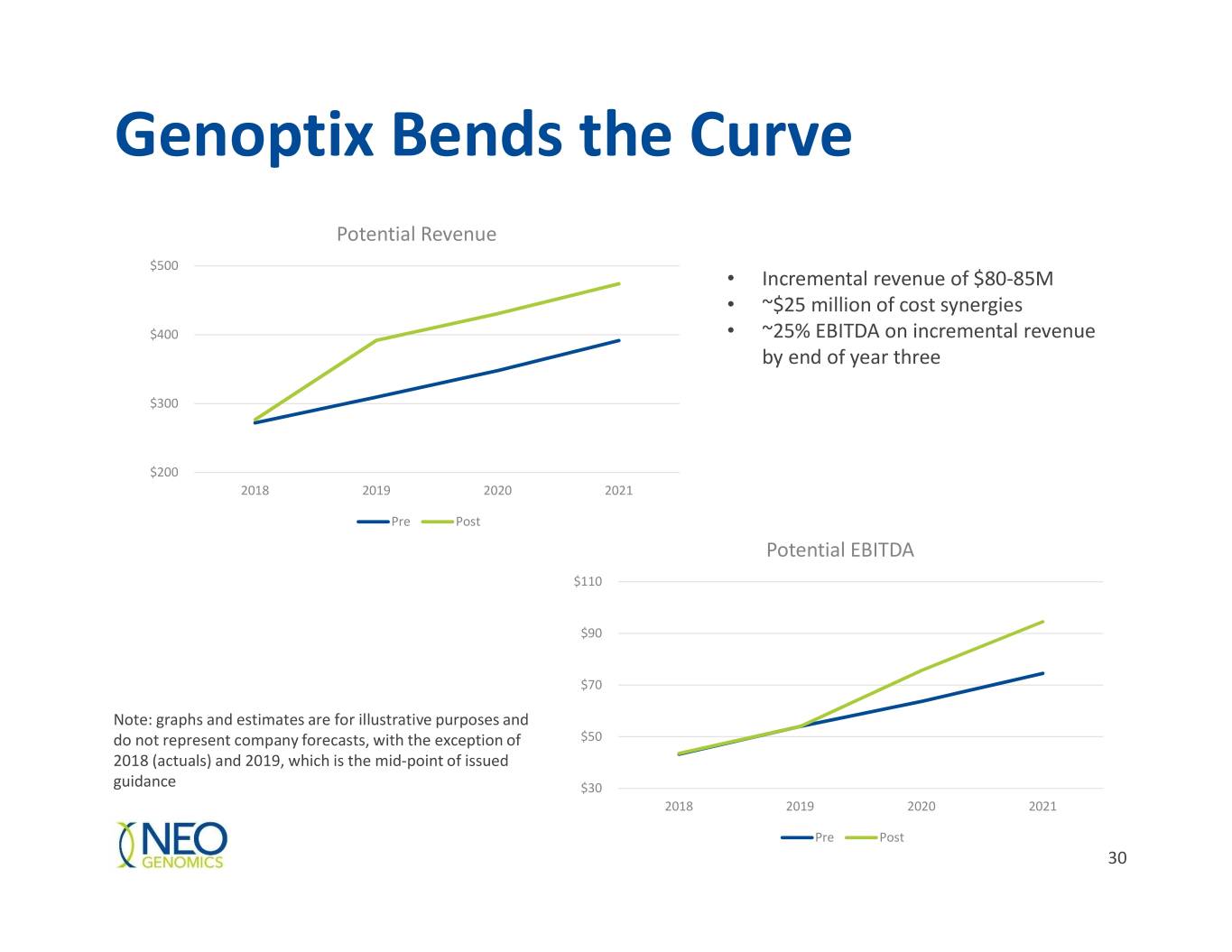
Genoptix Bends the Curve Potential Revenue $500 • Incremental revenue of $80-85M • ~$25 million of cost synergies $400 • ~25% EBITDA on incremental revenue by end of year three $300 $200 2018 2019 2020 2021 Pre Post Potential EBITDA $110 $90 $70 Note: graphs and estimates are for illustrative purposes and do not represent company forecasts, with the exception of $50 2018 (actuals) and 2019, which is the mid-point of issued guidance $30 2018 2019 2020 2021 Pre Post 30
